Sucampo and Abbott Announce Launch of AMITIZA in Japan
| CAS number | 136790-76-6 |
|---|
Sucampo Pharmaceuticals, Inc. and Abbott today announced the availability of AMITIZA® (lubiprostone) in Japan, a prescription medicine for the treatment of chronic constipation not caused by organic diseases. AMITIZA was approved by the Ministry of Health, Labor and Welfare (MHLW) in Japan in June.
“Chronic constipation is a medical condition affecting several million people in Japan and is one of the most common digestive condition complaints. Healthcare providers will now have a prescription option to help treat this condition more effectively and longer term, compared to currently approved over-the-counter medications”
“Millions of patients in Japan have been suffering from chronic constipation, without efficacious and well-tolerated, long-term treatment options. We are pleased to bring AMITIZA’s experience of six years and over six million prescriptions in the United States to the Japanese market, to benefit the healthcare providers and patients who need it,” says Ryuji Ueno, M.D., Ph.D., Ph.D., Sucampo’s Chairman and Chief Executive Officer.
“Chronic constipation is a medical condition affecting several million people in Japan and is one of the most common digestive condition complaints. Healthcare providers will now have a prescription option to help treat this condition more effectively and longer term, compared to currently approved over-the-counter medications,” says Akihiko Honda, General Manager, Abbott Japan.
AMITIZA may significantly advance the treatment of chronic constipation and will be the only prescription option available in Japan for this condition. The medicine is the world’s first chloride channel activator approved for therapeutic use, and has a unique mode of action that induces natural bowel movement frequency that is more in line with normal bowel habits.
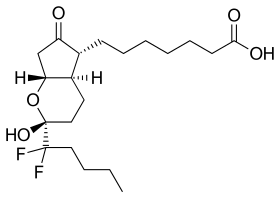
7-[(1R,3R,6R,7R)-3-(1,1-difluoropentyl)-3-hydroxy-
8-oxo-2-oxabicyclo[4.3.0]non-7-yl]heptanoic acid

Lubiprostone (rINN, marketed under the trade name Amitiza) is a medicationused in the management of chronic idiopathic constipation and irritable bowel syndrome. It was approved by the U.S. Food and Drug Administration (FDA) for this purpose on 31 January 2006.
Lubiprostone is used for the treatment of chronic constipation of unknown cause and irritable bowel syndrome associated with constipation.[1]
As of 20 July 2006, Lubiprostone had not been studied in children. There is current research underway to determine the efficacy in postoperative bowel dysfunction, and opioid-induced bowel dysfunction.

Sobrera, L. A.; Castaner, J. (2004). Drugs of the Future 29 (4): 336.