Sanofi Receives Positive CHMP Opinion in the European Union for Once-Daily Lyxumia® (lixisenatide)
 Sanofi announced today that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) has issued a positive opinion recommending the approval of once-daily Lyxumia® (lixisenatide) for the treatment of adults with type 2 diabetes mellitus to achieve glycemic control in combination with oral glucose-lowering medicinal products and/or basal insulin when these, together with diet and exercise, do not provide adequate glycemic control.
Sanofi announced today that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) has issued a positive opinion recommending the approval of once-daily Lyxumia® (lixisenatide) for the treatment of adults with type 2 diabetes mellitus to achieve glycemic control in combination with oral glucose-lowering medicinal products and/or basal insulin when these, together with diet and exercise, do not provide adequate glycemic control.
Lixisenatide is a once-daily injectable GLP-1 receptor agonist that has been developed by Sanofi.[1] As of September 2010 it is in clinical trials for diabetes.[2]
Lixisenatide, a glucagon-like peptide-1 agonist (GLP-1), is in development for the treatment of patients with type 2 diabetes mellitus. Lixisenatide was discovered by Zealand Pharma A/S and the global rights are licensed to Sanofi. Lyxumia® is the intended trademark for lixisenatide. Lixisenatide is not currently approved or licensed anywhere in the world.
GLP-1 is a naturally-occurring peptide that is released within minutes of eating a meal. It is known to suppress glucagon secretion from pancreatic alpha cells and stimulate insulin secretion by pancreatic beta cells. GLP-1 receptor agonists are in development as an add-on treatment for type 2 diabetes and their use is endorsed by the European Association for the Study of Diabetes, the American Diabetes Association, the American Association of Clinical Endocrinologists and the American College of Endocrinology.
The GetGoal phase III clinical program will provide data for lixisenatide in adults with type 2 diabetes treated with various oral anti-diabetic agents or insulin. With ten trials in the program, GetGoal started in May 2008 and has enrolled more than 4,300 patients. To date, GetGoal-X, GetGoal-L, GetGoal-L Asia, GetGoal-Mono, GetGoal-S and GetGoal-F1 have reported positive top-line results supporting efficacy and safety for lixisenatide
- Christensen, M; Knop, FK; Holst, JJ; Vilsboll, T (2009). “Lixisenatide, a novel GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus”. IDrugs : the investigational drugs journal 12 (8): 503–13. PMID 19629885.
- http://www.genengnews.com/gen-news-highlights/positive-phase-iii-data-for-two-separate-type-2-diabetes-therapies-reported-at-esad/81243945/
Sanofi Submits Application for Regulatory Approval for Lyxumia
®
(lixisenatide) for the Treatment of Type 2 Diabetes in Japan
 http://en.sanofi.com/Images/30529_20120611_LYXUMIA-JAPAN_en.pdf
http://en.sanofi.com/Images/30529_20120611_LYXUMIA-JAPAN_en.pdf
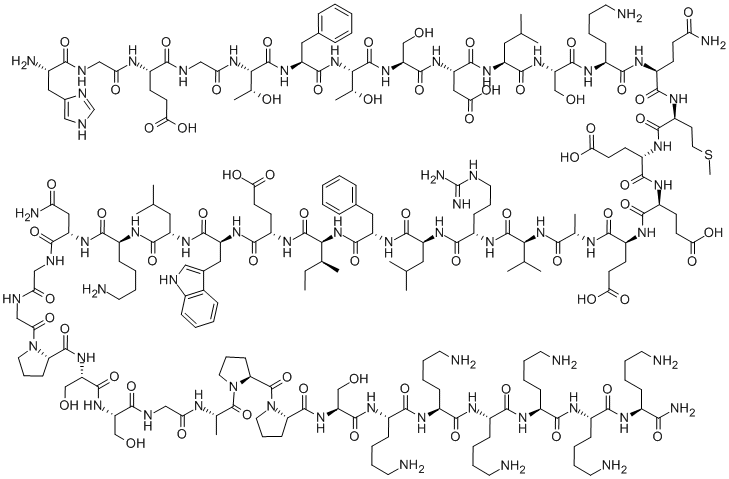
| CAS No. | 320367-13-3 | ||
| Chemical Name: | Lixisenatide | ||
| Synonyms: | lixisenatide | ||
| CBNumber: | CB01518519 | ||
Molecular Formula:
|
C215H347N61O65S |


str from chemblink Online Database of Chemicals from Around the World

 http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/002445/smops/Positive/human_smop_000448.jsp&mid=WC0b01ac058001d127
http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/002445/smops/Positive/human_smop_000448.jsp&mid=WC0b01ac058001d127



