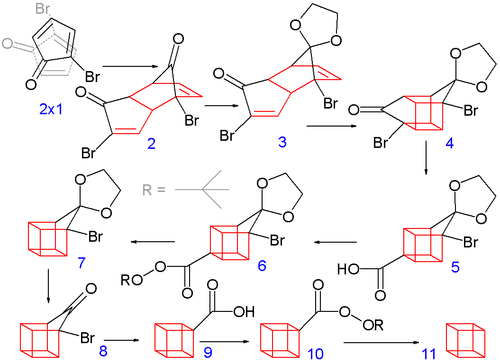
| CubaneCubane (C8H8) is a synthetic hydrocarbon molecule that consists of eight carbon atoms arranged at the corners of a cube, with one hydrogen atom attached to each carbon atom. A solid crystalline substance, cubane is one of the Platonic hydrocarbons. It was first synthesized in 1964 by Philip Eaton, a professor of chemistry at the University of Chicago. Before Eaton and Cole’s work, researchers believed that cubic carbon-based molecules could not exist, because the unusually sharp 90-degree bonding angle of the carbon atoms were expected to be too highly strained, and hence unstable. Once formed, cubane is quite kinetically stable, due to a lack of readily available decomposition paths | |
|---|---|
 |
 |

References :
| J. Am. Chem. Soc. 1964, 86, 962. (10.1021/ja01059a072) |
| J. Am. Chem. Soc. 1964, 86, 3157. (10.1021/ja01069a041) |

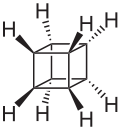
SYNTHESIS OF CUBANE

NBS
CCl4
wohl ziegler rxn